Join / Login > Class 11 > Chemistry > Structure of Atom. You will need to refer to a periodic table for proton values. Click hereto get an answer to your question From the symbol 19K40, state the atomic number of potassium. In this notation, the atomic number is not included. Symbol-mass format for the above atom would be written as Cr-52. For an example of this notation, look to the chromium atom shown below:Īnother way to refer to a specific atom is to write the mass number of the atom after the name, separated by a hyphen. The "A" value is written as a superscript while the "Z" value is written as a subscript. Both the atomic number and mass are written to the left of the chemical symbol. The composition of any atom can be illustrated with a shorthand notation called A/Z format. 
Additionally, potassium compounds have applications in medicine, agriculture, manufacturing, and energy storage, making potassium a versatile and valuable element.\nonumber \] Its role in maintaining fluid balance, nerve conduction, and muscle function underscores its significance in biological systems. In summary, potassium is a crucial element for the human body, agriculture, and various industries. Therefore, it is important to maintain a balanced intake of potassium and seek medical advice when necessary. It’s worth noting that while potassium is essential for bodily functions, excessive intake of potassium supplements or certain medical conditions can lead to high potassium levels (hyperkalemia), which can be potentially dangerous. 
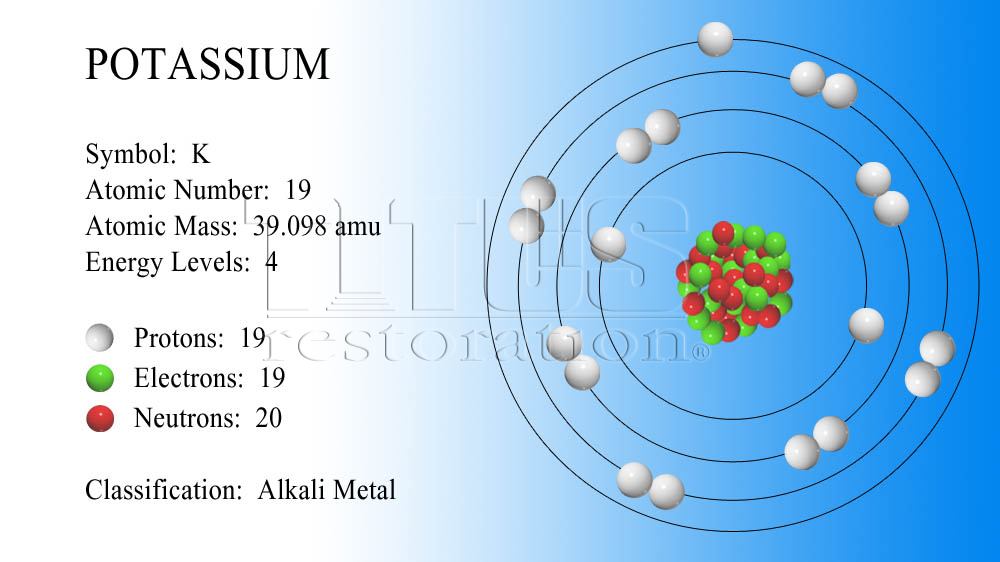
Potash, a term used to refer to potassium-rich minerals and compounds, is an important source of potassium for various industrial and agricultural purposes. Potassium-ion batteries utilize potassium ions instead of lithium ions for energy storage, offering potential advantages such as lower cost and improved safety.įurthermore, potassium is found in several minerals, such as potassium feldspar, which is a common component of many rocks. Potassium-ion batteries, a type of rechargeable battery, have been developed as a potential alternative to lithium-ion batteries. Energy levels 1 Electronegativity 2.20 Melting point -259.1. Potassium also has applications in energy storage. Potassium carbonate (K2CO3) is used in glass manufacturing, as a food additive, and in the production of fertilizers and other chemicals. Potassium compounds, like potassium hydroxide (KOH), are utilized in the production of soaps, detergents, and various chemicals. In addition to its biological and agricultural importance, potassium has industrial uses as well. Potassium promotes root growth, improves drought tolerance, and enhances overall plant health. Potassium-based fertilizers, such as potassium sulfate and potassium nitrate, are used to enrich the soil with potassium, which is essential for plant growth and development. Potassium also has agricultural applications. It can be administered orally or intravenously under medical supervision. Potassium chloride is commonly used as a supplement to correct potassium deficiencies in patients with certain medical conditions or in cases of low potassium levels (hypokalemia). Potassium compounds, such as potassium chloride, are used in medical settings. Potassium is obtained through dietary sources, and a balanced intake is crucial for overall health. It is an electrolyte that helps maintain proper electrical potentials and pH levels within cells. What are the valence electrons of potassium Potassium is an element of group-1 and it is an alkali metal. Therefore, the number of neutrons in potassium is 20. In the human body, potassium plays a vital role in maintaining fluid balance, conducting nerve impulses, and regulating muscle contractions, including the heartbeat. We know that the atomic number of potassium is 19 and the atomic mass number is about 39 (39.0983u). Potassium is an essential mineral for the human body and has several important functions. It is an alkali metal and is known for its highly reactive nature. Potassium is the nineteenth element on the periodic table with the chemical symbol K. Some interesting facts of Potassium are given below – Potassium has density ‘0.86’ and it is found ‘2.58’% on earth. Question : write some information about Potassium ?Īnswer : Potassium has melting point = 64 Question : write the electron configuration of Potassium element ?Īnswer : Potassium electronic configuration is ” 4s1”. Answer : as we know Potassium element is denoted by ‘K’ symbol and Potassium has ‘39.098’ atomic mass and ’19’ atomic number. Number of neutrons Mass numberAtomic number 99 43 56 Potassium 19 K L M N 2 8 8 1 Right electronic configuration is 2, 8, 6 X Y Proton 8 8. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
